Which of the Following Is a Lewis Base
Thus NH 3 is a Lewis base. Silver ion is a Lewis acid.
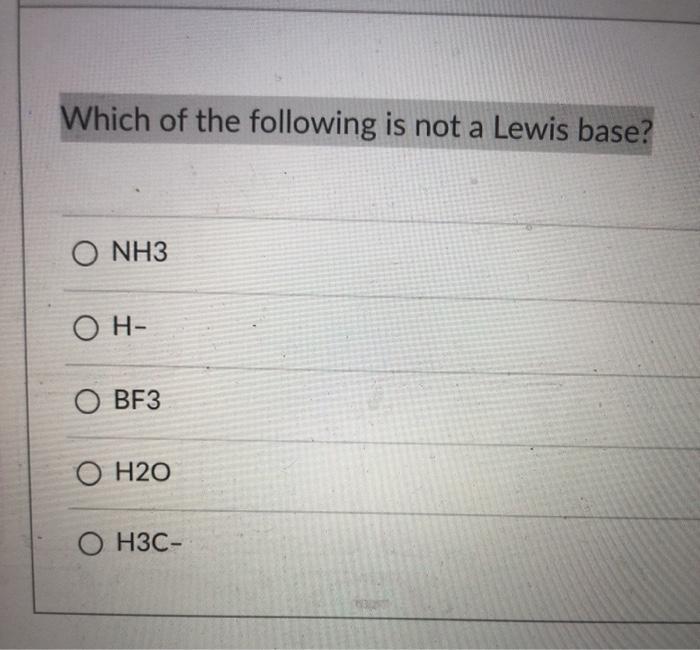
Solved Which Of The Following Is Not A Lewis Base O Nh3 On Chegg Com
Correct option is D CN ROH and NH 3.
. Which of the following is a Lewis base. The pH goes down. NH3aq Which of the following week be present in an aqueous solution of H2SO4.
The species which can donate a lone pair of electron are called Lewis base. The correct option is A. D Lewis bases are proton acceptors.
9 hours agoFerrari is still the strongest team on the grid after three Grands Prix. The reaction of Lewis acid-base can be represented by the transfer of pairing electrons to an acid medium from a base. In 1923 chemists Johannes Nicolaus Brønsted and Thomas Martin Lowry in their various studies created definitions of.
Which of the following can act as a Lewis acid. Which of the following is a Lewis base. NF 3 has no vacant orbital neither in nitrogen nor in fluorine so it cannot accept the electron and hence cannot acts as lewis acid and but for PCl 5 P has no LP hence it cannot acts as a base but ClF 3 3 BP 2 LP SF 4 4 BP 1 LP.
A compound that can acts as both a Brønsted -Lowry acid and base together are generally called amphoteric compounds. B magnesium chloride. At the beginning of the season the Italian team managed to build a car that seems to perform well on every circuit.
Which of the following is a Lewis base. Lewis concept does explain the behaviour of _____. A compound can be both Bronsted acid and Lewis base at the same time.
This states that a Lewis base is a nucleophile. While other molecules like BCl 3. C aluminium chloride D sodium ion View Answer Explanation.
A NH4OH B NaHCO3 C CaCO3 D KOH E NaO. To make it simple it is a substance through which a pair of electrons is donated to form a covalent bond. As a result hydrogen bonds with nitrogen atom.
Which statement about the following equilibrium is correct. Which of the following statements about Lewis bases is true. 2 A g 2 B g C g A 00058 atm B 0058 atm C 0076 atm D 0093 atm E 00076 atm.
Produces hydroxide ion in aqueous solution. 42Which one of the following is a true statement. Kp 19 x 102.
H_2 O C_5 H_12 AlF_3 SiF_4 None of the above are Lewis bases. A Lewis base is a donor of electron-pair. An element that has both nature is H2PO4-.
Is a strong electrophile and has vacant orbital to accept electron lone pair. The products will be favored because. Or if there is any difference at allSo for example is it better to use a boron trifluoride diethyl ether complex or a boron trifluoride tetrahydrofuran complex.
Class 11 Chemistry Mcqs. List the compound in marble that is destroyed by acid rain. HCl and Ag are have less electron density in their molecules so they cannot donate pair of electrons hence they are not Lewis base.
HSO4- aq NH3 aq SO42- aq NH4 aq the reactants will be favored because ammonia is a stronger base than the sulfate anion. Therefore when ammonia is dissolved in HCl it will accept the hydrogen atom as nitrogen gave electron to the hydrogen atom. H_2 O C_5 H_12 AlF_3 SiF_4 None of the above are Lewis bases.
Have lone pair of electrons and act as lewis bases. A AlF3 B H2O C SiF4 D C5H12 E None of the above are Lewis bases. Can be a substance that does not contain a hydrogen atom.
Chemistry questions and answers. Solve any question of Equilibrium with-. Carbon molecules which have pie electrons ie multiple bonds of a carbon have more electron density in that bond which can be easily donated to other species so they generally act as lewis base.
Thus it is a Lewis acid. Produces protons in aqueous solution. Agaq A Lewis acid.
According to Claudio Albertini this success is by no means a coincidence as he explained in conversation with The Race. Which of the following substances can act as an Arrhenius base a Brønsted-Lowry base and a Lewis base. Which of the following is a Lewis base.
The conjugate base of a strong acid is a strong base. The acid dissociation constant Ka equals 126 10-2 for HSO4- and is 56 10-10 for NH4. C Lewis bases are proton donors.
Which of the following is a Lewis base. D H2S E SİBr4 Calculate P Beq if P Aeq 04 atm P Ceq 035 atm for the following reaction. What is the effect on the pH when AlCl3aq is dissolved in water.
Lewis bases are electron pair donors. According to Lewis a base is a substance or specie which is able to donate electrons. A Lewis bases are electron pair acceptors.
B Lewis bases are electron pair donors. Strong acids can have negative pKa values. The stronger the acid the larger is its pKa.
Which of the following is a Lewis base. Eg NH 3 H 2 O Cl - etc. A AlCl3 B C6H14 None of the above is a Lewis base.
For example is a lewis base because nitrogen has a lone pair of electron. I am trying to understand Lewis bases bear with me I am a physicist. School MCQs Sub Category.
I was asking myself how diethyl ether and tetrahydrofuran compare with respect to their properties as solvents in electrophilic additions. Hello following is not a Lewis base it will talk about a space for the dial the species which which donate donate electron pairs so 2020 base right here so here we are having the sea and -2 veg and NH3 light show in D3 if you look that is having - 6 Jan similarly it is having lone pair of electrons so they will act as Lewis they will act as Lewis base plate and if we talk about alcl3. Acid-base reactions always favor the formation of the stronger acid and the stronger base.
Which of the following compound CANNOT act as a Lewis base.

Classify Each Of The Following As A Lewis Acid Or A Lewis Base Explain How To Determine Lewis Acids And Bases Home Work Help Learn Cbse Forum
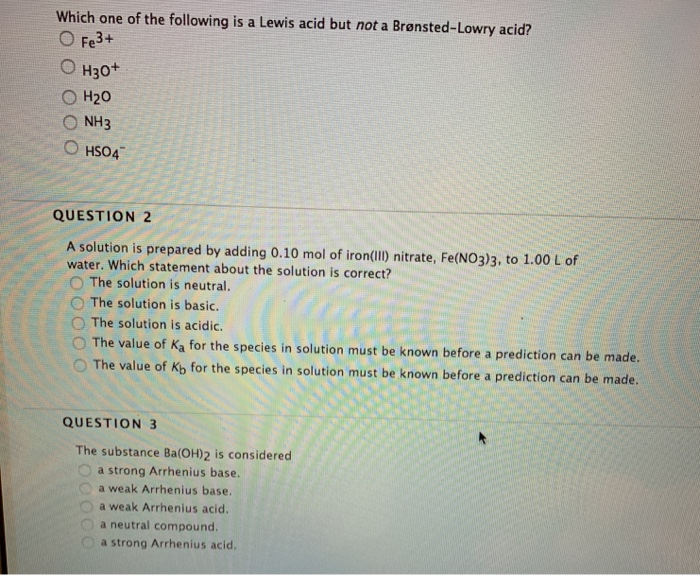
Solved Which One Of The Following Is A Lewis Acid But Not A Chegg Com

Which Of The Following Is Not A Lewis Base Youtube
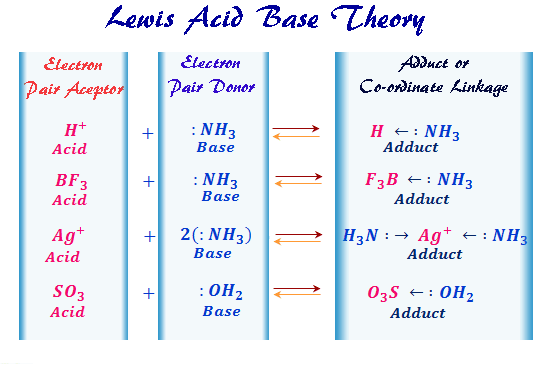
Lewis Acids Bases Definition Theory Properties Examples
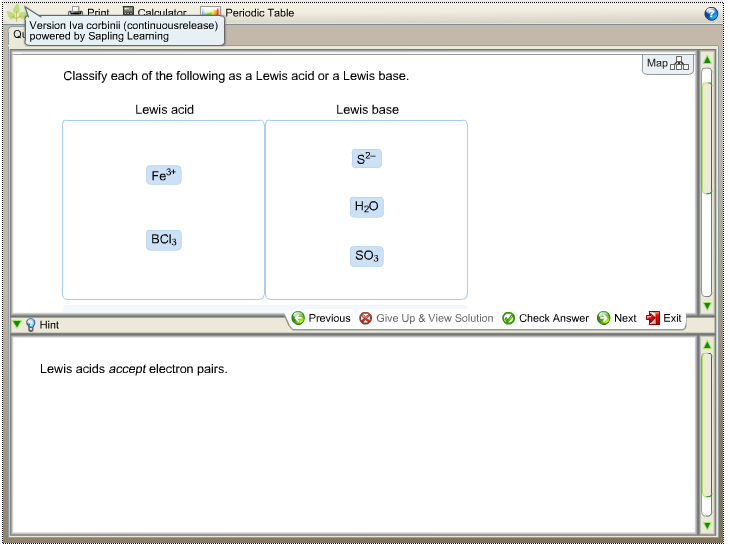
Solved Classify Each Of The Following As A Lewis Acid Or A Chegg Com
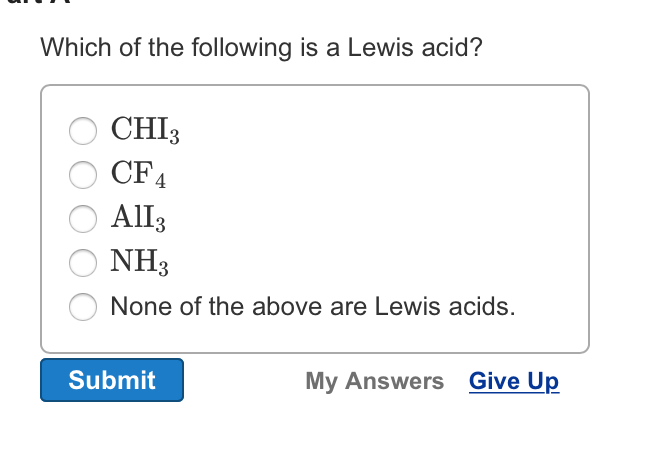
Solved Which Of The Following Is A Lewis Acid Chi 3 Cf 4 Chegg Com

Lewis Acids And Bases Chemistry Steps

Which Of The Following Is A Lewis Acid But Not A Bronsted Acid Youtube

Solved 6 Which Of The Following Is A Lewis Acid A All3 Chegg Com

Solved Which Of The Following Is A Lewis Base H 2 O C 5 Chegg Com

Solved Which Of The Following Is A Lewis Acid H O Bf 3 Chegg Com
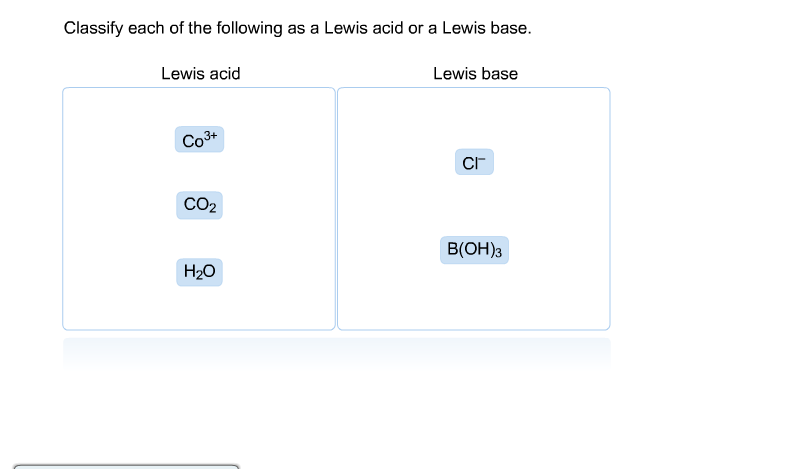
Solved Of The Following As A Lewis Acid Or A Lewis Base Chegg Com

Lewis Acids And Bases Definition Properties Examples Reactions Uses Applications Of Lewis Acids And Bases
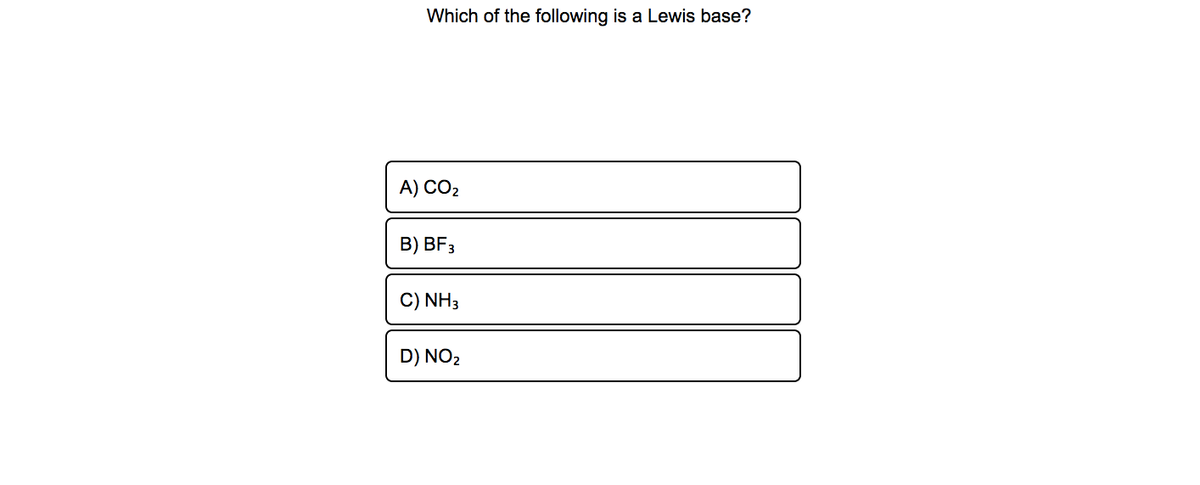
Answered Which Of The Following Is A Lewis Base Bartleby

Which Of The Following Is Lewis Base Youtube

Which Of The Following Is A Lewis Acid Youtube

Solved Question 16 Which Of The Following Is A Lewis Acid Chegg Com
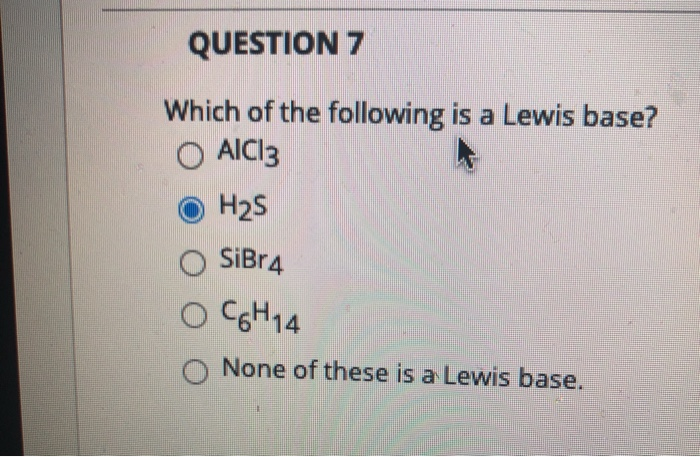
Solved Question 7 Which Of The Following Is A Lewis Base O Chegg Com
Comments
Post a Comment